DBV Aims to Unlock the Powerful Immune Properties of the Skin
Our investigational therapeutic treatment is based on epicutaneous immunotherapy, or EPIT. This potential new class of immunotherapy is designed to deliver microgram amounts of allergens to the immune system through intact skin using our proprietary VIASKIN® patch technology.
In addition to food allergies, the VIASKIN® patch technology is also being investigated as a treatment option for other immunological disorders. Additional applications for the VIASKIN® patch, such as non-invasive method for delivering vaccines against certain diseases, are also being investigated.
Merging Science and Technology
The information contained in this video is under clinical investigation. Products utilizing the VIASKIN® patch technology are for investigational use only and have not been approved by the FDA, or by any other regulatory authority, for any use.
Proposed Mechanism of Action of the VIASKIN® Patch in Food Allergy
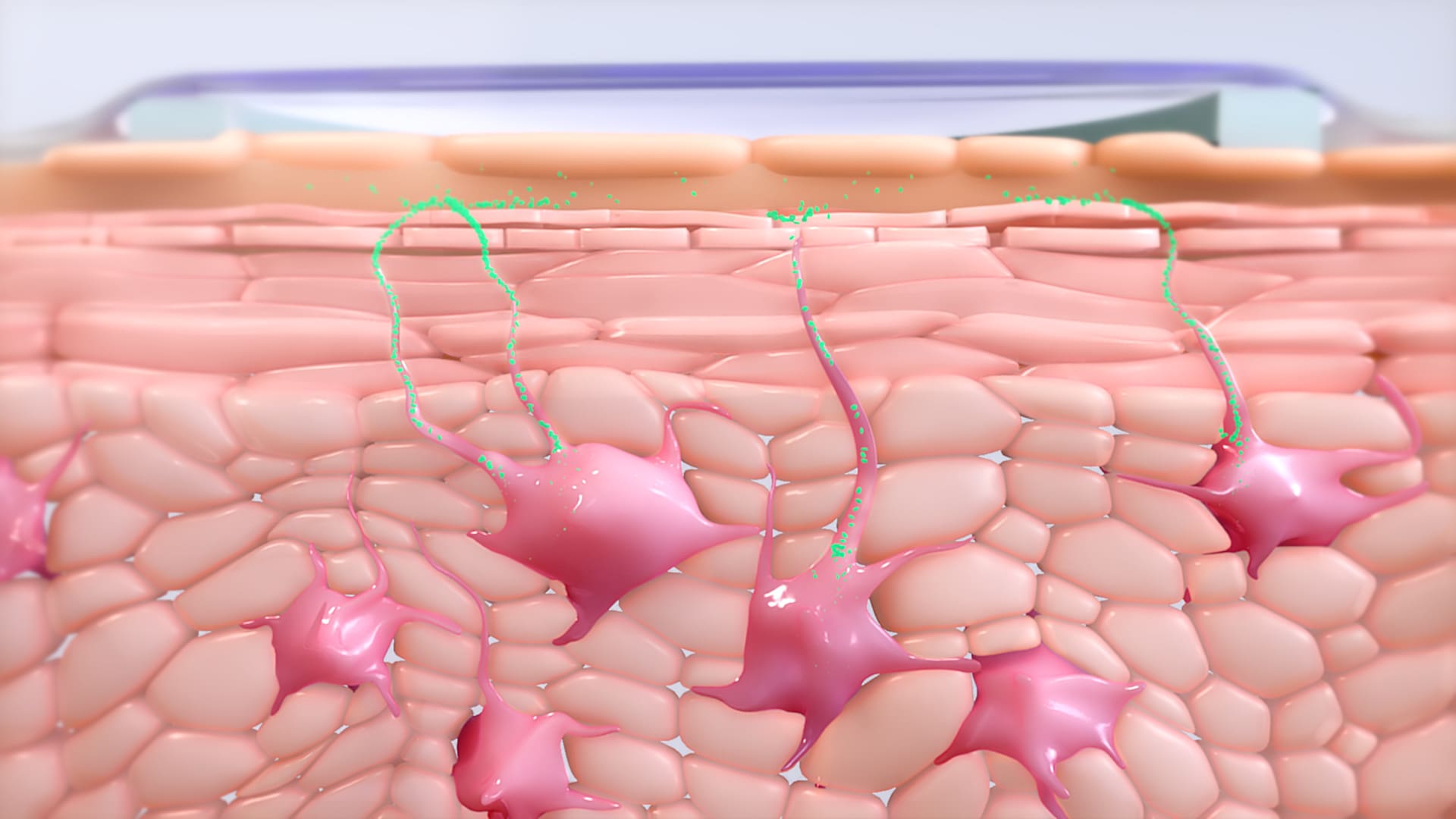
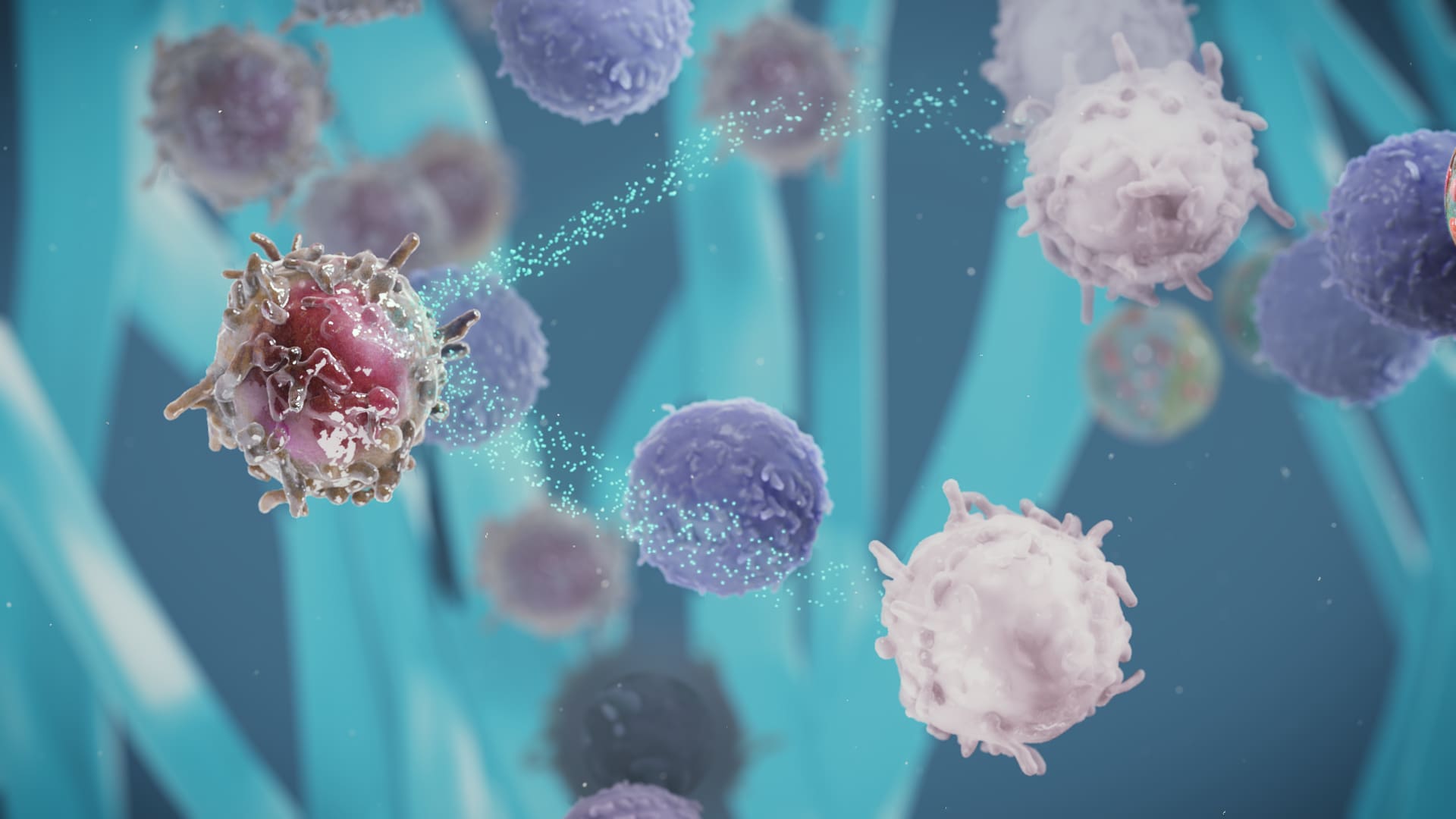
The skin’s ability to promote a tolerogenic immune profile in response to allergen exposure makes it a promising route for allergen immunotherapy. Our investigational approach to EPIT aims to target specific cells in the epidermis, such as Langerhans cells, in order to activate the immune system. Pre-clinical research has shown that these cells capture allergens and migrate to the lymph node where they activate specific regulatory T cells (Tregs). The specific Tregs activated by EPIT with the VIASKIN® patch can down-regulate the Th2-oriented reaction to the allergen, which leads to suppression of an allergic response.
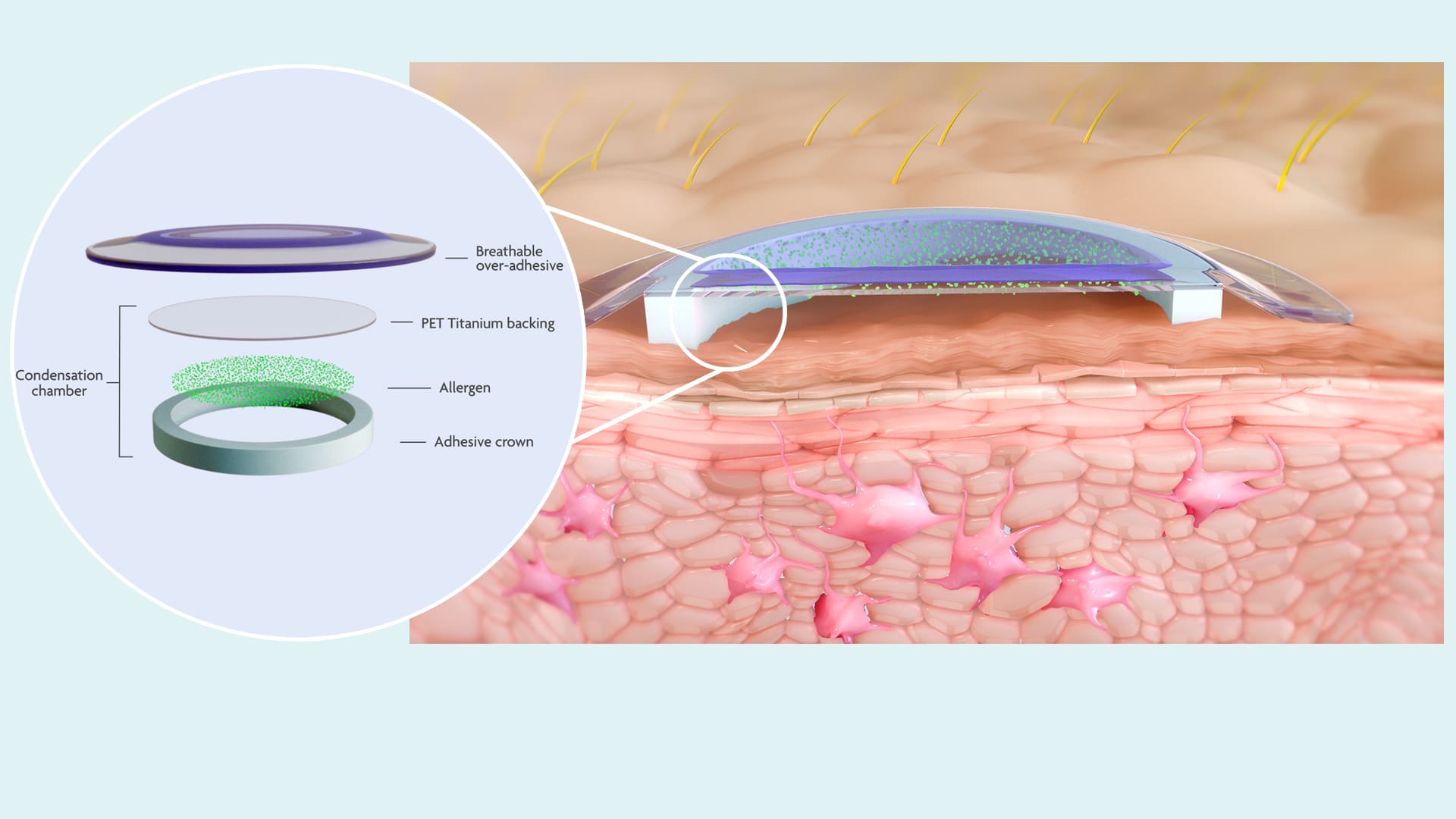
Key Elements of the VIASKIN® Patch’s Mechanism of Action
The VIASKIN® patch contains a deposit of dry allergen at its center, that sits above the skin on a backing film. When the patch is applied to intact skin, a condensation chamber is formed between the allergen and the top layer of skin, the epidermis.
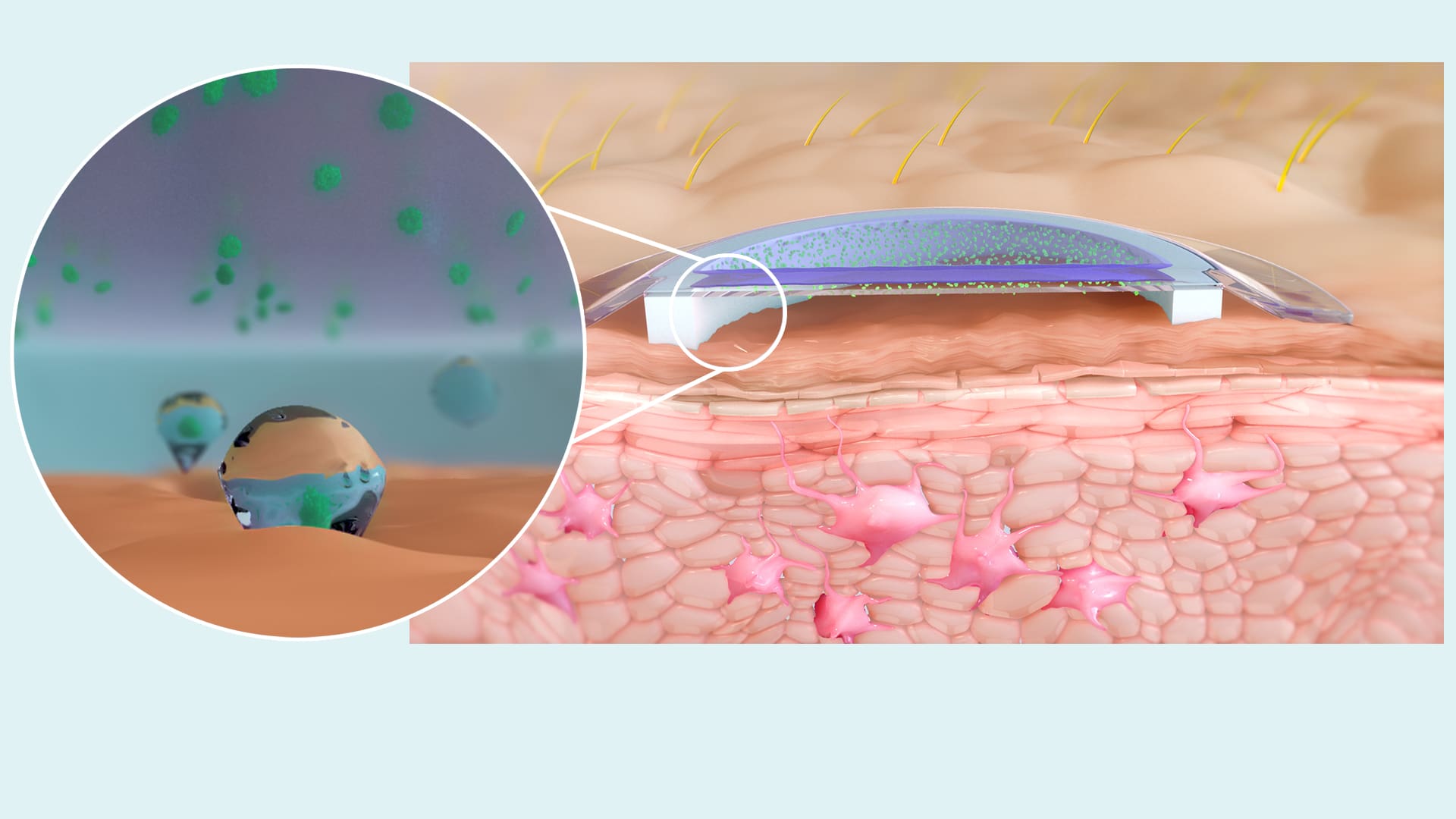
Within the epidermis, the allergen is captured by antigen-presenting cells, such as Langerhans cells (LC). Based on animal studies of EPIT with the VIASKIN® patch, there is no penetration of the allergen beyond the epidermis, limiting its access to the bloodstream.